Researchers at Oregon State University (OSU) have discovered a sustainable alternative to the ubiquitous Lithium-ion battery. Potassium-ion batteries, thought to be a technological dead-end for the last 83 years, could be made from much more abundant chemical components at cheaper prices.
The last time the possibility of K-ion batteries was looked into was 1932 and the scientists were obviously unsuccessful at that time. "For decades, people have assumed that potassium couldn’t work with graphite or other bulk carbon anodes in a battery," said Xiulei Ji, assistant professor of chemistry at OSU. "That assumption is incorrect," asserts Ji. "It's really shocking that no one ever reported on this issue for 83 years."
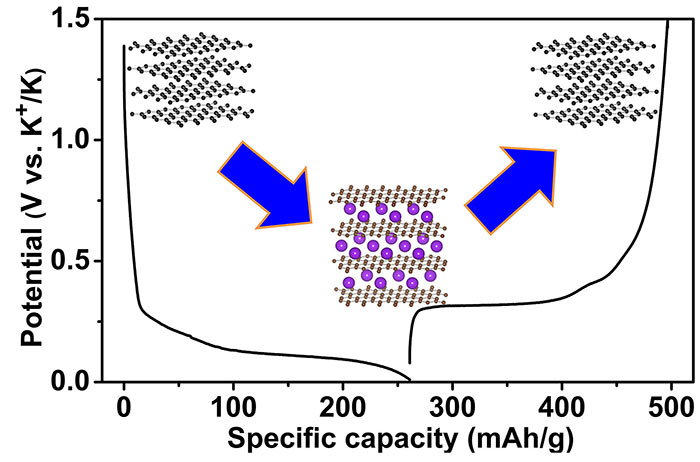
The researchers say their new success with K-ion batteries could be very important to the industry as they can work with well-established and inexpensive graphite as the anode. Lithium is a rare element, found in just 0.0017 percent of the earth's crust, by weight, and it is difficult to recycle. Potassium is 880 times more common.
Currently potassium ion batteries don't offer the energy density of Li-ion batteries, and the scientists don't think potassium will ever be able to power cells better than lithium. However with development the performance should get close to lithium technology batteries with a long cycling life, a high power density, plus a lot lower costs.
The OSU team are looking to do further research and at commercialisation options. It is envisaged that K-ion batteries will be used, like Li-ion batteries in devices such as smartphones and computers, but also in transportation, industry power backup, and micro-grid storage for home generated renewable energy.













